Introduction
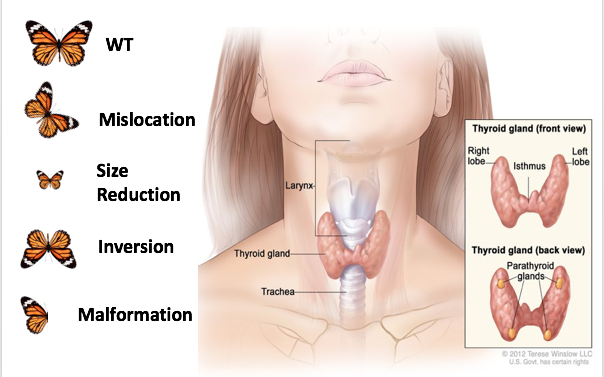
Congenital hypothyroidism (CH) is an autosomal recessive genetic disorder that disrupts the development of he thyroid gland and endocrine system. (1) Approximately 1:3000 newborns are diagnosed with CH, majority of which being women at a rate 3x more compared to men. The only treatment options for those with CH is daily hormone replacement therapy. The PAX8 gene is responsible development of the thyroid gland and thyroid stimulating hormone receptor (TSHR). Without the TSHR thyroid hormones production is compromised halting overall growth and development of individuals, disrupt metabolism, and left untreated results in learning disabilities. Mutations in PAX8 that result in multiple thyroid gland phenotypes such as malformations, size reductions, and mislocations are all association with CH. (2) [Fig.1] Each phenotype directly correlates to CH, yet no research has been conducted identifying the PAX8 genotypes for each thyroid gland phenotypes of those with CH. (3,4)[Fig.2]
I hypothesized that specific PAX8 genotypes correlates to each thyroid gland phenotype associated with CH.
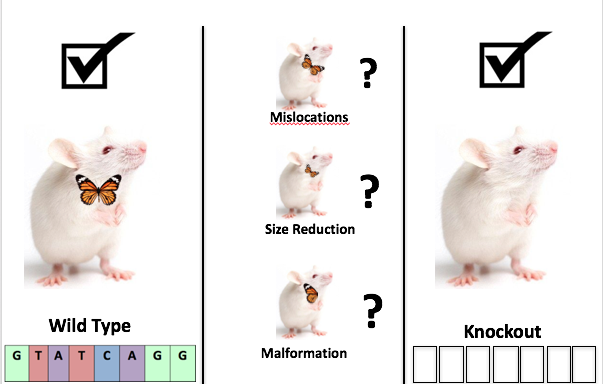
My primary goal is to Investigate thyroid gland genotypes and identify specific nonsynonymous mutations associated with each phenotype (mislocation, reduce size, malformation) within the coding regions of PAX8. In order to address my goal and answer my hypothesis I will be using the first Identify each genotype for all thyroid gland phenotypes, then Induce the mutant genotypes into wildtype (WT) models and finally find the Interruptions within the PAX8 to TSHR pathway.
I hypothesized that specific PAX8 genotypes correlates to each thyroid gland phenotype associated with CH.
My primary goal is to Investigate thyroid gland genotypes and identify specific nonsynonymous mutations associated with each phenotype (mislocation, reduce size, malformation) within the coding regions of PAX8. In order to address my goal and answer my hypothesis I will be using the first Identify each genotype for all thyroid gland phenotypes, then Induce the mutant genotypes into wildtype (WT) models and finally find the Interruptions within the PAX8 to TSHR pathway.
Specific Aim 1
|
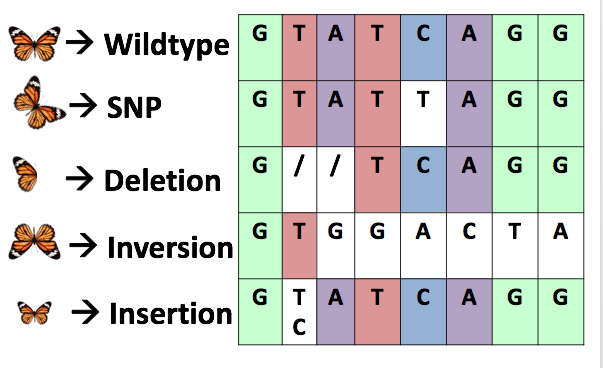
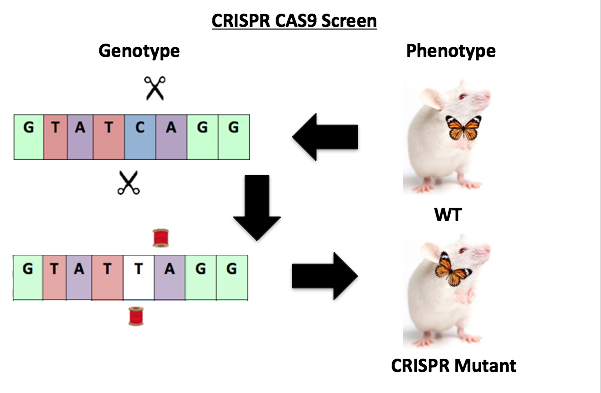
The first step is to Identify each of the PAX8 genotypes and pin point the mutations within the gene that result in each individual thyroid gland phenotypes of CH. Thyroid gland tissue are collected from mice models that represent all the known mutant phenotypes (malformation, size reduction, mislocation, ect) and using Next Generation Sequencing and CLUSTAL Omega to create consensus sequence of each mutant PAX8 genotypes that correspond to each thyroid gland phenotype of CH. [Fig. 3]
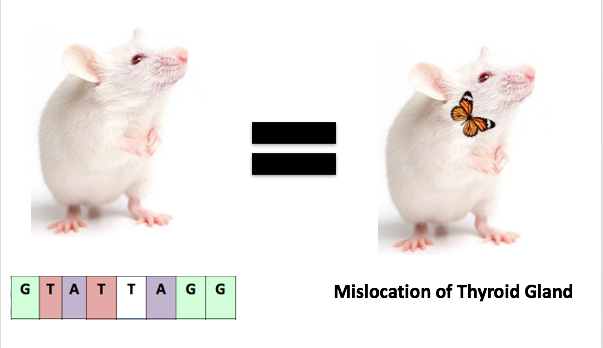
I hypothesize that specific mutant PAX8 genotype correspond to each of the thyroid gland phenotypes. As shown in Fig. 4, if provided a mouse or individual with an unknown phenotype, but a known PAX8 mutant genotype one could infer the phenotype. |
Fig. 3
Fig. 4
|
Specific Aim 2
|
Fig. 5
Fig. 6
|
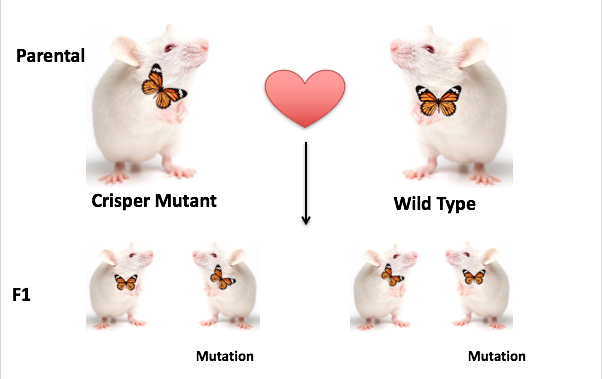
Now to double check whether the consensus sequences for each mutant genotypes yields the desires mutant thyroid gland phenotype we will Induce all PAX8 mutant genotypes in mice models. Using a CRISPR- CAS9 Screen WT mice will be subjected to the introduction of a known PAX8 mutant genotypes that is associated with a specific thyroid gland phenotype (malformation, size reduction, ect). [Fig. 5] The CRISPR mutant mice will be used to answer whether thyroid gland phenotypes associated with CH are heritable. Crossing CRISPR Mutant mice to WT and investigating the offspring enhanced insight on the who PAX8 mutant genotypes are inherited. CH being a recessive autosomal genetic disease the first step to understanding it is to study how PAX8 mutations are transmitted from parent to offspring. [Fig. 6]
I hypothesize that mutant parental mice with an induced PAX8 genotype that results in a mislocation thyroid gland phenotype, the offspring of the mutant parental mice will inherit (1:3) both genotype and phenotype of parent . |
Specific Aim 3
|
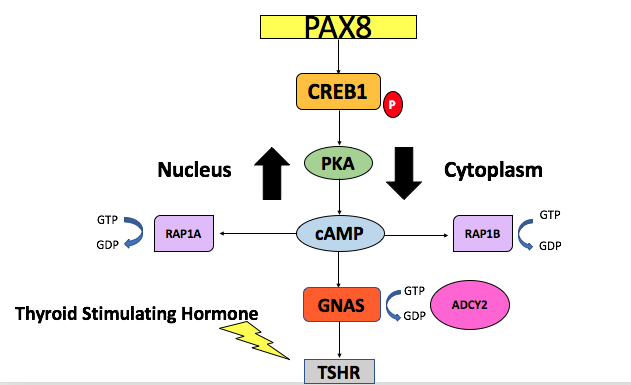
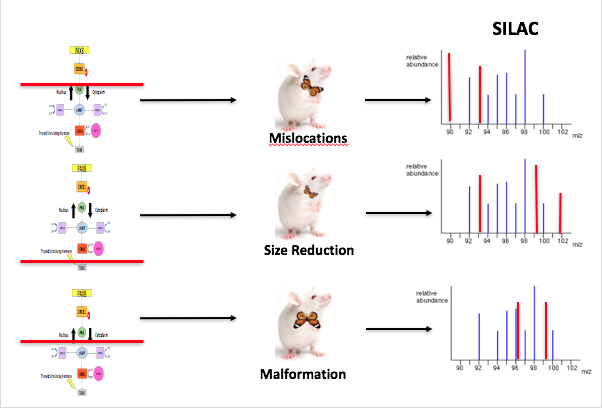
Lastly we will investigate how protein interactions differ in the PAX8 to TSHR pathway for each thyroid gland phenotype. PAX8 is the first step in the TSHR cascade, the intermediate steps in between are a series of phosphorylation events activating each protein all the way down to TSHR. [Fig. 7] Using SILAC Mass Spectroscopy (MS) will enable us to determine the biological interactions of PAX8 and TSHR proteins from mice model representing each thyroid gland phenotypes associated with CH.
I hypothesize that within the PAX8 and TSHR pathway intermediate proteins localize in the thyroid gland at different quantities and abundance compared to WT mice model. [Fig. 8 ] Identifying the interruptions of PAX8 and TSHR may provide insight on development of CH per thyroid gland phenotype. |
Fig. 7
Fig. 8
|
Future Directions
The newborn screening process is comprised of a blood panel that is tested for an array endocrine related disease, one of which is CH. However, current methods of screening are antiquated and if a positive test arises no phenotype nor genotype is provided. Technicians with a positive CH blood test called out to the newborns physician to diagnose the child with CH. Parents with children with CH often see specialist known as Endocrinologist several times a year especially in the earliest years of life. These visits are expensive and it can take a few visits to identify a newborns thyroid gland phenotype and determine dosage hormone therapy as early as within the first week of life.
This study could provide the necessary preliminary data to create an assay used in during the newborn screening that provides instant knowledge of a newborns thyroid gland phenotype. [Fig. 9] Inventing such assay could potentially save physicians time and parents money, but more importantly newborns with CH could have excess to more accurate dosage and faster hormone replacement treatments.
This study could provide the necessary preliminary data to create an assay used in during the newborn screening that provides instant knowledge of a newborns thyroid gland phenotype. [Fig. 9] Inventing such assay could potentially save physicians time and parents money, but more importantly newborns with CH could have excess to more accurate dosage and faster hormone replacement treatments.
Fig. 9
References:
[1]Magliano, M. Pasca Di, R. Di Lauro, and M. Zannini. "Pax8 has a key role in thyroid cell differentiation." Proceedings of the National Academy of Sciences 97.24 (2000): 13144-3149.
[2]Park, S. M. "Genetics of congenital hypothyroidism." Journal of Medical Genetics 42.5 (2005): 379-89.
[3]Ramos, H. E., et al. "Extreme phenotypic variability of thyroid dysgenesis in six new cases of congenital hypothyroidism due to PAX8 gene loss-of-function mutations." European Journal of Endocrinology 171.4 (2014): 499-507
[4]Tell Gianluca, et al. "Structural defects of a Pax8 mutant that give rise to congenital hypothyroidism." Biochemical Journal 341.1 (1999): 89.
[2]Park, S. M. "Genetics of congenital hypothyroidism." Journal of Medical Genetics 42.5 (2005): 379-89.
[3]Ramos, H. E., et al. "Extreme phenotypic variability of thyroid dysgenesis in six new cases of congenital hypothyroidism due to PAX8 gene loss-of-function mutations." European Journal of Endocrinology 171.4 (2014): 499-507
[4]Tell Gianluca, et al. "Structural defects of a Pax8 mutant that give rise to congenital hypothyroidism." Biochemical Journal 341.1 (1999): 89.
| simard_564genrough_2_21_pp_copy.pptx | |
| File Size: | 3388 kb |
| File Type: | pptx |
| simard_564gen_pax8_pp.pdf.zip | |
| File Size: | 5071 kb |
| File Type: | zip |